Publications
University of North Carolina at Chapel Hill
Honey KL, Torzone SK, Dowen RH. The C. elegans flr-3(ut9) mutation is a loss-of-function insertion within the drl-1 locus. microPublication Biology 2023 ![]()
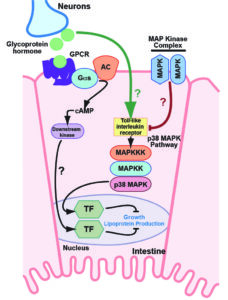 Torzone SK, Park AY, Breen PC, Cohen NR, Dowen RH. Opposing action of the FLR-2 glycoprotein hormone and DRL-1/FLR-4 MAP kinases balance p38-mediated growth and lipid homeostasis in C. elegans. PLoS Biology 2023 Sep 29;21(9):e3002320. PMID: 37773960
Torzone SK, Park AY, Breen PC, Cohen NR, Dowen RH. Opposing action of the FLR-2 glycoprotein hormone and DRL-1/FLR-4 MAP kinases balance p38-mediated growth and lipid homeostasis in C. elegans. PLoS Biology 2023 Sep 29;21(9):e3002320. PMID: 37773960
Animals integrate developmental and nutritional signals before committing crucial resources to growth and reproduction; however, the pathways that perceive and respond to these inputs remain poorly understood. Here, we demonstrate that DRL-1 and FLR-4, which share similarity with mammalian mitogen-activated protein kinases, maintain lipid homeostasis in the C. elegans intestine. DRL-1 and FLR-4 function in a protein complex at the plasma membrane to promote development, as mutations in drl-1 or flr-4 confer slow growth, small body size, and impaired lipid homeostasis. To identify factors that oppose DRL-1/FLR-4, we performed a forward genetic screen for suppressors of the drl-1 mutant phenotypes and identified mutations in flr-2 and fshr-1, which encode the orthologues of follicle stimulating hormone and its putative G protein-coupled receptor, respectively. In the absence of DRL-1/FLR-4, neuronal FLR-2 acts through intestinal FSHR-1 and protein kinase A signaling to restrict growth. Furthermore, we show that opposing signaling through DRL-1 and FLR-2 coordinates TIR-1 oligomerization, which modulates downstream p38/PMK-1 activity, lipid homeostasis, and development. Finally, we identify a surprising noncanonical role for the developmental transcription factor PHA-4/FOXA in the intestine where it restricts growth in response to impaired DRL-1 signaling. Our work uncovers a complex multi-tissue signaling network that converges on p38 signaling to maintain homeostasis during development. ![]()
Gurley NJ, Szymanski RA, Dowen RH, Butcher TA, Ishiyama N, Peifer M. Exploring the evolution and function of Canoe’s intrinsically disordered region in linking cell-cell junctions to the cytoskeleton during embryonic morphogenesis. PLoS One 2023 Aug 3;18(8):e0289224. PMID: 37535684
Dowen RH, Ahmed S. Maternal Inheritance: Longevity Programs Nourish Progeny via Yolk. Curr Biol 2019 Aug 5;29(15):R748-R751. PMID: 31386852 ![]()
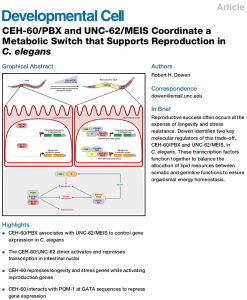
Dowen, RH. CEH-60/PBX and UNC-62/MEIS coordinate a metabolic switch that supports reproduction in C. elegans. Dev Cell 2019 Apr 22;49(2):235-50. PMID: 30956009
The molecular basis of how animals integrate metabolic, developmental, and environmental information before committing resources to reproduction is an unresolved issue in developmental biology. In C. elegans, adult animals reallocate fat stores from intestinal cells to the germline via low-density lipoprotein (LDL)-like particles to promote embryogenesis. Here, I demonstrate that two conserved homeodomain transcription factors, CEH-60/PBX and UNC-62/MEIS, coordinate a transcriptional network that supports reproduction while suppressing longevity and stress response pathways. The CEH-60:UNC-62 heterodimer serves an unanticipated dual function in intestinal nuclei by directly activating the expression of lipoprotein genes while directly repressing stress-responsive genes. Consequently, ceh-60 mutants display fat storage defects, a dramatic lifespan extension, and hyper-activation of innate immunity genes. Finally, CEH-60 associates with PQM-1 at the DAF-16-associated element within the promoters of stress-responsive genes to control gene expression. Thus, CEH-60 governs an elaborate transcriptional network that balances stress responses and longevity against reproduction during developmental transitions. ![]()
Massachusetts General Hospital
Massachusetts General Hospital
Dowen, RH, Breen, PC, Tullius, T, Conery, AL, Ruvkun, G. A microRNA program in the C. elegans hypodermis couples to intestinal mTORC2/PQM-1 signaling to modulate fat transport. Genes Dev 2016 Jul 1;30(13):1515-28. PMID: 27401555 ![]()
Cheesman, HK, Feinbaum, RL, Thekkiniath, J, Dowen, RH, Conery, AL, Pukkila-Worley, R. Aberrant Activation of p38 MAP Kinase-Dependent Innate Immune Responses Is Toxic to Caenorhabditis elegans. G3 2016 Jan 27;6(3):541-9. PMID: 26818074
Riedel, CG, Dowen, RH, Lourenco, GF, Kirienko, NV, Heimbucher, T, West, JA, Bowman, SK, Kingston, RE, Dillin, A, Asara, JM, and Ruvkun, G. DAF-16 employs the chromatin remodeller SWI/SNF to promote stress resistance and longevity. Nat Cell Biol 2013 May;15(5):491-501. PMID: 23604319
Montgomery, TA, Rim, YS, Zhang, C, Dowen, RH, Phillips, CM, Fischer, SE, and Ruvkun, G. PIWI associated siRNAs and piRNAs specifically require the Caenorhabditis elegans HEN1 ortholog henn-1. PLoS Genet 2012 Apr 19;8(4). PMID: 22536158
University of California San Diego
Qi, D, Dubiella, U, Kim, SH, Sloss, DI, Dowen, RH, Dixon, JE, and Innes, RW. Recognition of the protein kinase AVRPPHB SUSCEPTIBLE1 by the disease resistance protein RESISTANCE TO PSEUDOMONAS SYRINGAE5 is dependent on s-acylation and an exposed loop in AVRPPHB SUSCEPTIBLE1. Plant Physiol 2014 Jan;164(1):340-51. PMID: 24225654
Dowen, RH, Pelizzola, M, Schmitz, RJ, Lister, R, Dowen, JM, Nery, JR, Dixon, JE, and Ecker, JR. Widespread dynamic DNA methylation in response to biotic stress. Proc Natl Acad Sci USA 2012 Aug 7;109(32):E2183-91. PMID: 22733782
Lister, R*, Pelizzola, M*, Dowen, RH, Hawkins, RD, Hon, G, Tonti-Filippini, J, Nery, JR, Lee, L, Ye, Z, Ngo, Q, Edsall, L, Antosiewicz-Bourget, J, Stewart, R, Ruotti, V, Millar, AH, Thomson, JA, Ren, B, and Ecker, JR. Human DNA methylomes at base resolution show widespread epigenomic differences. Nature 2009 Nov 19;462(7271):315-322. *Equal contribution. PMID: 19829295
Dowen, RH, Engel, JL, Shao, F, Ecker, JR, and Dixon, JE. A Family of Bacterial Cysteine Protease Type III effectors utilize acylation-dependent and independent strategies to localize to plasma membranes. J Biol Chem 2009 Jun 5;284(23):15867-79. PMID: 19346252
Gentry, MS, Dowen, RH, Worby, CA, Mattoo, S, Ecker, JR, and Dixon, JE. The phosphatase laforin crosses evolutionary boundaries and links carbohydrate metabolism to neuronal disease. J Cell Biol 2007 Jul 23;178(3):477-88. PMID: 17646401
Digicaylioglu, M, Kaul, M, Fletcher, L, Dowen, R, and Lipton, SA. Erythropoietin protects cerebrocortical neurons from HIV-1/gp120-induced damage. Neuroreport 2004 Apr 9;15(5):761-3. PMID: 15073510